Metallurgy

This article does not have any sources. (September 2013) |
Metallurgy is the study of metals. Alloys (mixtures of metals) are also studied. There are two main branches of Metallurgy. They are physical and chemical metallurgy. A Metallurgist is individual who researches, manufactures and develops metal items that range from car parts to semiconductors.
Metallurgists and Metallurgical Engineers often work for consulting engineering firms; mining and manufacturing companies; and in government and universities. Many metallurgists hold a Bachelor of Engineering or similar 4- year degree.
Common Job Tasks
- Study the properties metals and alloys through scientific experiments
- Plan and design machines and processes to concentrate, extract, refine and process metals.[1]
- Work in a manner that is environmentally and economically conscious.
Chemical Metallurgy[change | change source]
Chemical Metallurgy is the study of extracting and refining metals through chemical reactions. This field also commonly involves corrosion prevention and surface treatment of metals. Scientific concepts such as thermodynamics, the study of heat flow and kinetics, the study of the speed of reactions are very central to chemical metallurgy.
Metallurgists are very interested in the chemical reactions that occur with different elements. Chemical reactions are carried out on a large scale to obtain a useful product.
For example, the production of steel in a blast furnace involves turning different Iron and oxide compounds into pure Iron.[2]
Physical Metallurgy[change | change source]
Physical metallurgy is the science of making useful properties out of metals. This field focus on the physical property of metals and alloys such as strength and hardness. Properties of metals might be electrical, magnetic and physical in nature. These properties can be changed by alloying or heat treatment.
There are many ways to make metals. For example, large parts may be created by pouring liquid metal into a large cast in a process called casting. [3]
Important factors in physical metallurgy include time and temperature in which the metal is made and cooled. Metals undergo different phase changes, the particular position and type of crystal structure inside the metal will determine if a metal is brittle or flexible. [4]
Smelting[change | change source]
Ores are rocks that have a chemical compound of a metal with oxygen, called an oxide. Separating the oxygen from the metal is called smelting. This is done with chemistry or electricity, usually at very high temperatures. This is the first step in metallurgy. A rock containing enough metal to be profitable is called ore.
Alloys[change | change source]
An alloy is a combination of a metal with one or more elements. The metal mixture must be a solid to be called as an alloy. A common way to combine metals into an alloy is by melting them, mixing and then allowing them to solidify by cooling at room temperature. Alloys have been known since 50,000 BCE and have been continuously studied and improved.
Common Example of alloys include:
- Chromium alloys: Good at withstanding high temperature and have good resistance to corrosion. Common composition include a mixture of chromium and Nickel
- Cast Iron: Often used in making pots and pans. Cast Iron is a mixture of Iron and around 1-3% Carbon.[5] It is often made using a blast furnace.
- Bronze: Copper with 12-15% Tin and minor additions of other metals such as manganese, aluminum and zinc. Bronze is a common metal found in archeological sites.[6]
Metal parts[change | change source]
Another part of metallurgy is making parts from metals. These parts must be made so they will not break when they are used. Metallurgists look to make metal that meets the needs of the process it will be used in. Sometimes the metal must be lightweight or low in density. Other times it must be tough (not easily broken). The metallurgist must follow directions when making the part to know what metal to use. the process of metal selection is known as the materials selection process. Metallurgists use mathematical equations to determine what are the most important physical features of the metal. Common desired characteristics include ductility, toughness, corrosion resistance, and strength. Different metals and their associated features are often graphed on Ashby plots.[7]
A metal starts as a block, called an ingot. Metallurgists must know how to make a metal part from an ingot. Parts are made from ingots different ways. When a big hammer is used, it is called forging. To make thin metals, a metal is put between two rolls and moved, called rolling.
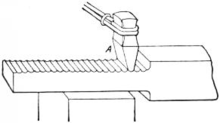
Making metal hot makes it easier to change the shape of a metal part. For this reason, most metal parts are made using hot metal. This is hot work.
Two metal parts can be put together with much heat. This is called welding. Iron is easy to weld.
Tools[change | change source]
Metallurgists use many tools to determine the characterizes of metal using a technique called metallography. One of the most useful tools is the Scanning Electron Microscope. To use an SEM a metal sample is polished and hit with electrons to produce a clear image of the metal. This permits for the metal microstructure to be seen. A microstructure is what the metal looks like when zoomed in very closely.[8] There are many types of microstructure such as pearlite and ferrite.
Metals are sometimes pulled until they break using a tensiometer. This is the tension test. Information such as plastic and elastic deformation can be determined from these tests.
It is also common for metallurgists to use computer simulations to predict heat flow or the cost of making a particular product. Common software used in metallurgy include ABAQUS[9] and ANSYS.[10]
References[change | change source]
- ↑ "Metallurgical and materials engineers". WorkBC. July 14, 2020. Archived from the original on May 23, 2022. Retrieved November 26, 2020.
- ↑ "23.3: Metallurgy of Iron and Steel". Chemistry LibreTexts. 2015-01-18. Archived from the original on 2021-05-10. Retrieved 2020-11-27.
- ↑ Kaye, Alan; Street, Arthur (1982). Die Casting Metallurgy | ScienceDirect. Butterworth Scientific. ISBN 9780408107174. Archived from the original on 2020-12-05. Retrieved 2020-11-27.
- ↑ Callister, William (2009). Materials Science and Engineering: An Introduction Hardcover – Dec 30 2009. Wiley. ISBN 978-0470419977.
- ↑ Campbell, F. C. (Flake C. ) (2008). Elements of metallurgy and engineering alloys. Library Genesis. Materials Park, Ohio : ASM International. ISBN 978-0-87170-867-0.
- ↑ Radivojević, Miljana; Roberts, Benjamin W.; Pernicka, Ernst; Stos-Gale, Zofia; Martinón-Torres, Marcos; Rehren, Thilo; Bray, Peter; Brandherm, Dirk; Ling, Johan (2019-06-01). "The Provenance, Use, and Circulation of Metals in the European Bronze Age: The State of Debate". Journal of Archaeological Research. 27 (2): 131–185. doi:10.1007/s10814-018-9123-9. ISSN 1573-7756. S2CID 149947751. Archived from the original on 2022-05-23. Retrieved 2020-11-27.
- ↑ Skakov, Yu.A. (May 2000). "The Formation Sequence of Intermediate Phases in Mechanical Alloying of Binary Systems". Materials Science Forum. 343–346: 597–602. doi:10.4028/www.scientific.net/msf.343-346.597. ISSN 1662-9752. S2CID 135534957. Archived from the original on 2022-05-23. Retrieved 2020-11-27.
- ↑ "Crystal Microstructure - an overview | ScienceDirect Topics". www.sciencedirect.com. Archived from the original on 2022-05-23. Retrieved 2020-11-27.
- ↑ "ABAQUS Student Edition | 3DEXPERIENCE Edu". edu.3ds.com. Archived from the original on 2021-03-03. Retrieved 2021-02-17.
- ↑ "Engineering Simulation & 3D Design Software | Ansys". www.ansys.com. Archived from the original on 2021-02-17. Retrieved 2021-02-17.



